 At the time of returning to their original orbit, they leave this energy as electromagnetic radiation. When these atoms grasp the energy and move into a higher orbit, this theory refers to them as “excited” electrons. When the electrons are in orbit, they possess “constant energy.” In 1913, Danish physicist Niels Bohr proposed a planetary model, which states that electrons revolve about the nucleus just as the planets orbit the sun. He further hypothesized that the number of protons and electrons are equals in an atom. In the past, he discovered the part of activity such as the movement of protons and electrons within the central part of the atom. In 1911, Ernest Rutherford (British physicist) proposed a nuclear model on atoms. Today we call the positive charged particles protons and the negative one’s electrons. He does so after discovering electrons in 1897.Īlso, his model suggested that atoms consist of a big positively-charged sphere studded with negatively charged electrons (he called them “corpuscles”) like fruit in a plum pudding.įurthermore, he put forward that the charge of the positive sphere’s charge is equal to the negative charges of the electrons. Thomson proposes the “plum pudding” theory of the divisible atom. He also proposed that while all atoms of one element are identical, they are totally different from those that make up other elements. He postulated that matter is made of atoms, which are small indivisible particles. Dalton’s Atomic TheoryĮnglish chemist John Dalton subsequently made on the Greek notion of atoms in 1808. Moreover, they also made some intangible qualities such as taste and color. In addition, they tell that these were solid particles without internal structure, and came in a variety of shapes and sizes. “All the matter is made up of tiny units called atoms” this was first proposed by Leucippus and Democritus, in the fifth century B.C., that all matter is made of tiny units called atoms. 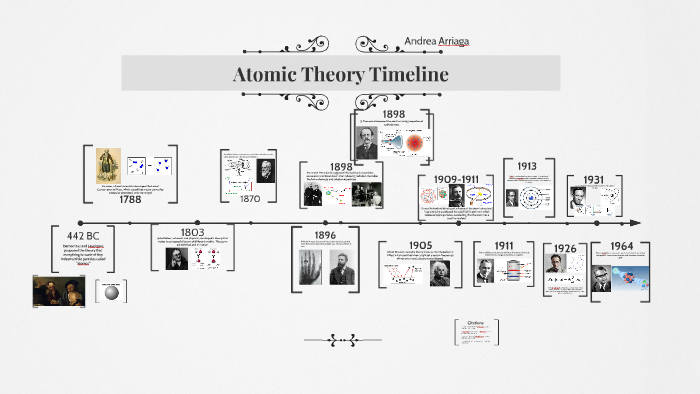
This was later found to be too specific a model and was pushed aside for the current theory of quantum mechanics which is based on mathematics and describes a probability of “locating” an electron.List of Atomic Theories 1. Ancient Greek Beliefs Niels Bohr suggested that electrons move on orbits like planets around a sun.Ernest Rutherford later showed that most of the mass and all of the positive charge of an atom is found in a small dense core of the atom that we call the nucleus.JJ Thomson is credited with proving the first part of Dalton’s theory incorrect when he discovered the electron, as a result of his cathode ray tube experiment.His theory included the ideas that atoms make up all matter, are indivisible, and that atoms of an element are identical. John Dalton completed scientific experiments to develop his atomic model with evidence.He is also credited with using the word " atomos" to describe a small unit of matter that was unbreakable. Democritus is often credited with the first atomic theory, though it was a philosophical idea without evidence. 
You can add more than one template to this assignment to give students lots of options! Suggested Scientists 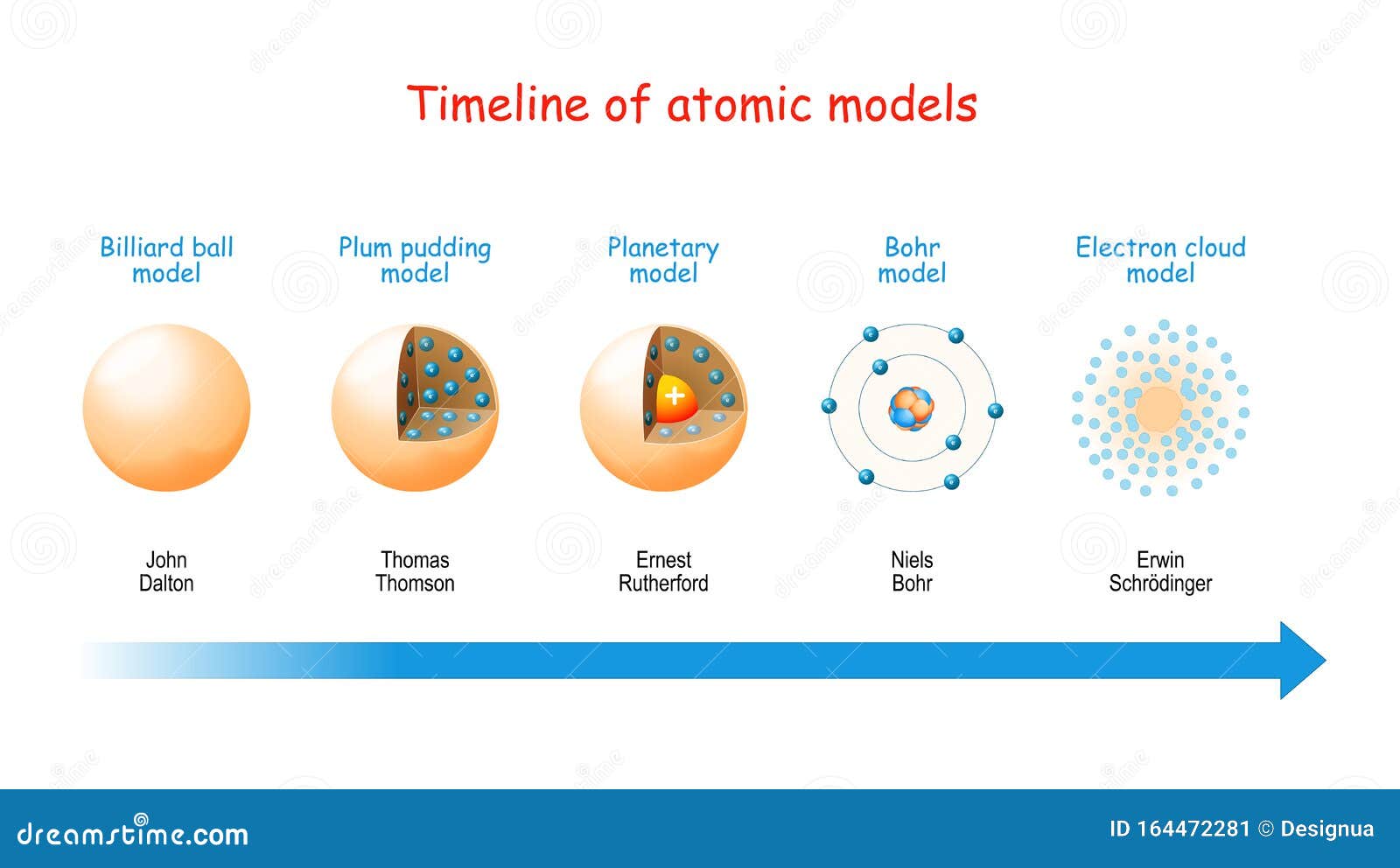
They can also describe the major advancements in each description box to make as detailed or general a timeline as is required by the level of chemistry they are studying.įor an alternative to the timeline layout, have students create a timeline poster to incorporate into a presentation or gallery walk. 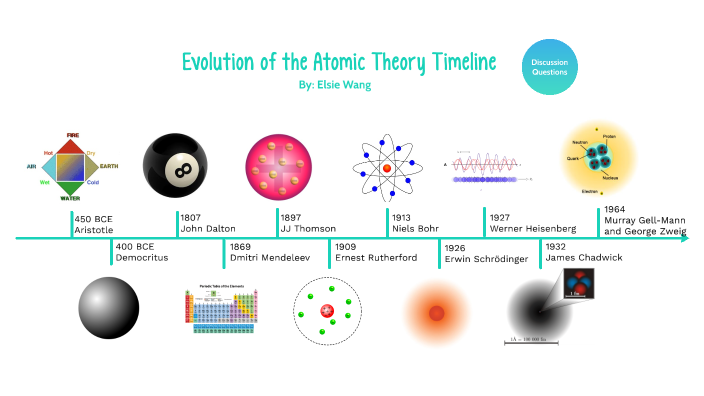
Students can upload pictures of these scientists or, in some cases, of their equipment, using Photos for Class. They should be sure to include a variety of different scientists and discoveries related to atomic theory in their timeline. Using a timeline layout, students will illustrate major atomic developments and outline how ideas changed over time. It is a valuable experience for students to understand that scientific ideas evolve over time in response to new and better data. Many lessons on atomic structure begin with a recitation of the historic developments that led to the current theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
